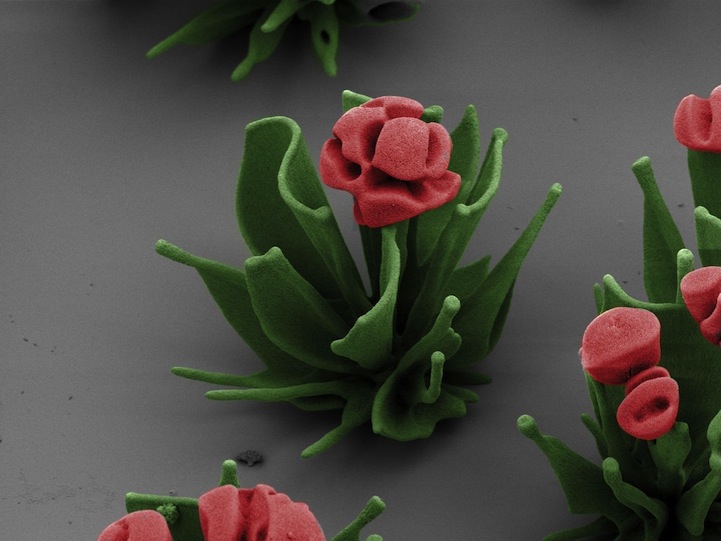
Harvard School of Engineering and Applied Sciences (SEAS) post-doctoral fellow Wim L. Noorduin has carefully manipulated chemical compounds to create microscopic structures that resemble flowers. That's not to say that he sculpted on a microscopic level, but rather combined solubles that naturally react with one another and the environment to result in self-assembled floral architecture.
“To create the flower structures,” Harvard SEAS explains, “Noorduin and his colleagues dissolve barium chloride (a salt) and sodium silicate (also known as waterglass) into a beaker of water. Carbon dioxide from air naturally dissolves in the water, setting off a reaction which precipitates barium carbonate crystals. As a byproduct, it also lowers the pH of the solution immediately surrounding the crystals, which then triggers a reaction with the dissolved waterglass. This second reaction adds a layer of silica to the growing structures, uses up the acid from the solution, and allows the formation of barium carbonate crystals to continue.”
Noorduin's process of manipulating chemical gradients is reflective of the development of any multi-celled organism. The scientist says, “For at least 200 years, people have been intrigued by how complex shapes could have evolved in nature. This work helps to demonstrate what's possible just through environmental, chemical changes.” The fields of self-assembled flowers present the results of precise changes like altering pH gradients to create “broad, radiating leaves, a thin stem, or a rosette of petals.”
Noorduin's research is featured on the cover of this month's issue of Science.












via [Visual News, Science Mag, Harvard SEAS]