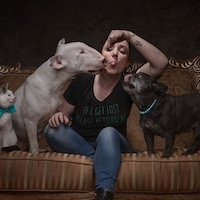Several patients who volunteered as subjects in the drug trial. (Photo: screenshot from a video by Memorial Sloan Kettering)
Cancer is one of the scariest six-letter words in the world. For decades, scientists and doctors have striven for a cure. Everyday people have run, walked, biked, and boated for a cure. In February 2022, President Biden even announced the re-ignition of the cancer moonshot to end the disease as we know it. Any victory in the war against cancer is worth celebrating. A new paper in The New England Journal of Medicine announced a shocking success to celebrate: in a drug trial of 18 subjects with rectal cancer, the cancer was eliminated in all 18 people.
Two doctors at Memorial Sloan Kettering Cancer Center had a question: what effect would using immunotherapy with a checkpoint inhibitor early in the stages of cancer have? Would it be more effective than using this drug treatment in later stages? Dr. Andrea Cercek and Dr. Luis A. Diaz Jr. wanted to find out. They began approaching drug companies, asking then to fund a clinical trial using checkpoint inhibitors to treat patients with locally advanced rectal cancer. This means the cancer is largely still confined to one area rather than spread out. Eventually, they found a company that was willing to take the risk. Tesaro, which was later bought by GlaxoSmithKline, agreed to fund the small study.
The doctors then recruited cancer patients to join the study. They first found Sascha Roth, an otherwise healthy 38-year-old who had recently been diagnosed with a rectal cancer. Her particular mutation made chemotherapy a less promising treatment, so she entered the drug trial. Others soon joined, totaling 18 individuals. They each took dostarlimab, a checkpoint inhibitor which helps the immune system identify and destroy cancer cells. They took the drug (which costs about $11,000 per dose) every three weeks for six months.
The results were shocking and even historic. The cancer was eliminated in all the patients, becoming undetectable by physical exam, endoscopy, PET scans, or MRI scans. “I believe this is the first time this has happened in the history of cancer,” Dr. Diaz said, referring to each study subject going into complete remission. While this is amazing news, the study results must be replicated before the effectiveness of the drug on certain patients can be fully understood. “Very little is known about the duration of time needed to find out whether a clinical complete response to dostarlimab equates to cure,” said Dr. Hanna K. Sanoff of the University of North Carolina’s Lineberger Comprehensive Cancer Center, who was not involved in the study. While this study is not announcing a cure, it is an exciting sign for treatments to come.
18 rectal cancer patients participated in a drug trial, and they all went into remission.
h/t: [IFL Science, Science Alert, The New York Times]
Related Articles:
Paralyzed Patients Walk and Swim Again After Receiving a Spinal Cord Implant
New Study Finds Ideal Amount of Sleep Needed as We Age
Scientists Have Completed Mapping the Last 8% Of the Human Genome
Researchers Discover That Your Brain’s Mental Speed Doesn’t Decline Until Your 60s