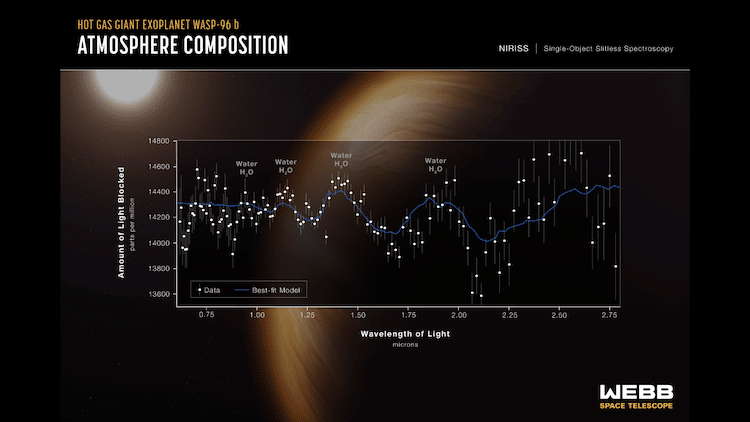
Spectroscopy of WASP 96-b's atmosphere (Photo: NASA)
There have been many incredible images released this month from the James Webb Space Telescope (JWST), showcasing how much has improved since Hubble. In addition to photos, there has also been some remarkable data released. Of all these dazzling findings, one of the most extraordinary discoveries is the detection of life on exoplanets.
Finding life outside of our own little world has been a never-ending journey for humankind. The first detection of exoplanets in the early ‘90s was confirmation that that journey could come to a conclusion in the near future. Exoplanets are planets outside our solar system that orbit their own star, much like the Earth orbits the Sun. There are now over 5,000 exoplanets that have been discovered by NASA, some of which could host life.
Though there is no magical telescope that can transmit hard proof of life on these planets that are light-years away, we do have JWST, which can measure the elemental makeup of the atmospheres of those planets. The presence of water or certain elements, such as oxygen, methane, and carbon dioxide—or, better yet, a combination of these—would indicate some existence of life. “If you, like, took some water and some methane and put them in a box and left them at room temperature on Earth, they’d actually combine into carbon dioxide,” ASU astronomer Megan Mansfield explains, “and you wouldn’t expect there to be any methane left over. The only reason we have methane in the Earth’s atmosphere is because it’s constantly produced by life.”
The process our powerhouse telescope uses to gather this information is known as the transit method. JWST is pointed in the direction of the exoplanet’s star. As the exoplanet travels between us and its star, the planet will block some of the starlight. The starlight that is not blocked will be filtered through the planet’s atmosphere and make it back to JWST, which then diffracts that light into a spectrum of colors. The data that is collected then gets plotted to create a spectrum, a type of graph that shows the intensity of light at different wavelengths.
Why a spectrum of colors? Certain elements and molecules absorb particular wavelengths of light. That light that is not absorbed then gets reflected back to the viewer, appearing as a certain color. For example, you will see leaves of dark green because they have a good amount of chlorophyll. Scientists know where certain elements lie in this grand spectrum of colors. In layman’s terms, JWST reveals what colors are staying in the planet’s atmosphere. Astronomers then decipher that data to determine what molecules are present in its climate and if there could be an indication of life.
This habitable exoplanet search is already underway, with Webb’s first reading in June 2022 of giant gas exoplanet WASP-96b. This gassy globe has temperatures of over 1000°F and is “much puffier than any planet orbiting our Sun.” Its spectrum proved a significant presence of water and, even more surprising, clouds and haze. On a planet as hot as WASP-96b, Cornell astrophysicist Nikole Lewis points out that rock could condense in the air like water can here on Earth. This means those clouds JWST detected could be made of sand. So, while this exoplanet’s size and temperature sadly disprove the idea of a habitable environment, we can all marvel at the idea that there is a planet out there where sand rains down from the clouds up above.
In the meantime, JWST will turn its sights onto other exoplanets. This includes the exoplanets orbiting the cool dwarf star TRAPPIST-1, specifically those in the star’s habitable zone. These could have liquid water, making them candidates for supporting life as we know it here on Earth. The James Webb Space Telescope will have an eventful journey of discovery and has already made its mark on our endless journey to find life out there in space.
The James Webb Space Telescope just proved it can detect signs of life on other planets by looking at the light that passes through their atmospheres.
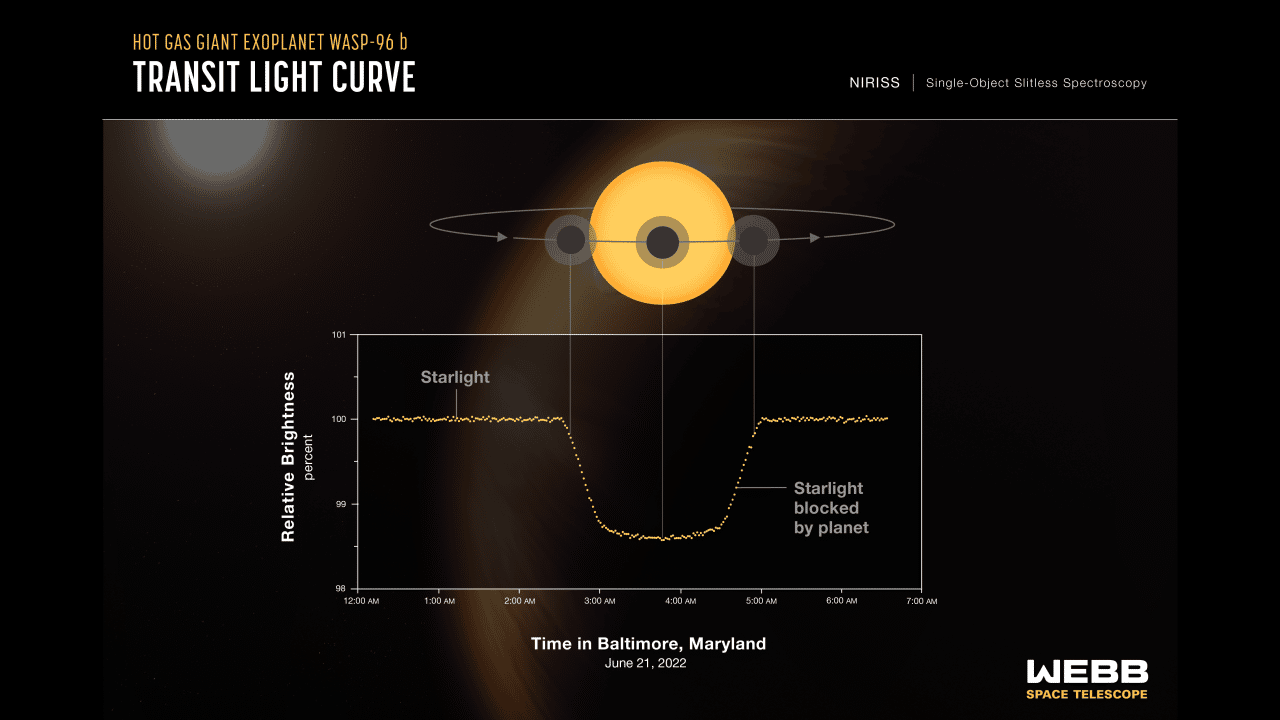
Spectroscopy showing WASP 96-b's transit light curve (Photo: NASA)
While the gas giant exoplanet WASP 96-b is presumably uninhabitable, JWST detected water and clouds in its skies. Those clouds could be made of sand.

Hypothetical Visualization of WASP 96-b (Photo: NASA)
Moving forward, the monumental telescope will set its sights on other exoplanets, like TRAPPIST-1e. If elements of life are out there somewhere, JWST will find them.

Artist Rendering of TRAPPIST-1e (Photo: NASA)
James Webb Space Telescope: Website | Facebook | Instagram | Twitter
h/t: [IFLScience]
Related Articles:
James Webb Space Telescope Has Photographed Jupiter, and the Results Are Incredible
James Webb Space Telescope Has Enough Fuel to Stay in Space for 20 Years
NASA Celebrates James Webb Space Telescope Successfully Reaching Its Destination